
Pharma Biotech Consultants
OUR INTEGRATED SERVICES
Clinical Development and Medical Affairs
Our extensive drug development expertise covers diversified therapeutic areas in a wide range of indications, spanning from phase I to phase IV, we:
- Translate strategic objectives into streamlined, actionable Global Development Plans (GDP)s
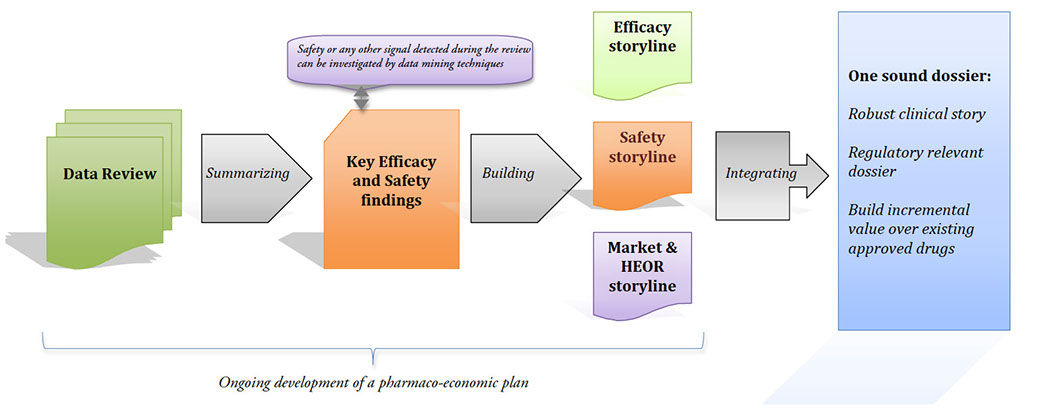
- Support the tactical execution of clinical trial programs: Clinical Development Plan (CDP), organize Steering Committees, organize and run Safety Monitoring Boards, prepare documents such as: Clinical Overview, Summary of Clinical Efficacy, Summary of Clinical Safety, etc…)
- Help with organizational design and operational process review, as well as building of clinical department
Strengthen diversified therapeutic franchises across all life cycle phases and maximize the commercial value of in-market products through new indications and line-extensions.
Pharmaco-Economics
Devise pharmaco-economic strategies (dossiers) to support Fair Market Value backing for pricing & reimbursement negotiations.
Design the appropriate PRO (Patient’s Research Outcome) tools, including appropriate quality of life scales in order to support pricing and reimbursement strategies.
Develop appropriate models with CHAD.
Develop and update analog research data to support value dossier.
Statistics
Define the best strategy to analyze and report data; this includes sophisticated analyses and graphs that will help understand and interpret the data supporting an indication.
Conduct complex epidemiological data analyses.
Business Development
Help guide product portfolio decisions: clinical and regulatory due diligence, market evaluation/ risk assessment of in-licensing opportunities.
Help conceive, develop and update Business Plans.
Regulatory Affairs
Extensive experience in interacting with major agencies across the world (FDA, Health Canada, EMA, Swiss Medic, PMDA, TGA, SFDA), we will help you:
- Define a global regulatory strategy and assemble the best submission package, devise core and region’s specific submission strategies
- Provide competitive and regulatory intelligence
- Prepare and facilitate regulatory authority meetings
- Share knowledge and insights on decision-making dynamics of major health regulatory agencies
- Help with organizational design and process review, as well as building of a regulatory affairs department
Drug Safety
Evaluate complex safety issues using a combination of traditional safety analyses and data mining associated with multidimensional space exploration:
- This innovative technique may identify several conditions (for example co-morbidities, predisposing factors, concomitant medications) that may play a contributing role to safety issues
- Based on findings, define the optimal strategy to address those issues
- Review and updates of scientific literature
- Design and or update Risk Management Plans